
Decarboxylation of acid yields a neutral substance (C), the nitration of which forms only one mono derivative (D). EXPLAINATION: Electron withdrawing groups like -NO2, increases the acidity. 
Hydrolysis of a compound A (C 7H 3Cl 5) gives an acid B of the formula C 7H 4Cl 2O 2. The decreasing order of their acidic strength: NO2CH2COOH > FCH2COOH > C6H5COOH. ❬OOH deactivates and directs incoming electrophilic to meta position. Halogenation is therefore the first step in the conversion of a carboxylic acid into many important substituted carboxylic acid. The halogen of these halogenated acid undergoes nucleophilic displacement and elimination same as it does in the simple alkyl halides. (v) CH 3COOH, Cl 2CHCOOH, CH 3CH 2COOH, Cl 3CCOOH, ClCH 2COOH (iv) ClCH 2COOH, CH 3CH 2COOH, ClCH 2CH 2COOH, (CH 3) 2CHCOOH,CH 3COOH (ii) CH 3COOH, (CH 3) 2CHCOOH, (CH 3) 3CCOOH Arrange the following illustration in order of increasing acidity (C) CH 3–CH 2–CHCl–COOH (D) CH 3CH 2–CCl 2 –COOHĬarboxylic acids are weak acids and their carboxylate anions are strong conjugate bases and are slightly alkaline due to the hydrolysis of carboxylate anion compared to other species, the order of acidity and basicity of corresponding conjugate bases are as follows:Īcidity RCOOH > HOH > ROH > HC º CH > NH 3 > RHĬH 3COOH + NaHCO 3 -> CH 3COO –Na + + H 2O + CO 2 (A) CH 2Cl–CH 2–CH 2COOH (B) CH 3CHCl –CH 2–COOH Which one of the following would be expected to be most highly ionised in water? (c) Increase in the electronegativity of halogen increases the acidity. This is due to the fact that inductive effect decreases with increasing distance. (b) Increase in the distance of Halogen from COOH decreases the acidity e.g. Included are several functional group equivalents considered to be at the same oxidation state. This is illustrated by the progression of a methyl group to a carboxylic acid in a series of 2-electron oxidations, as shown at right. (a) Increase in the number of Halogen atoms on a-position increases the acidity, eg.Ĭl 3CCOOH > Cl 2CHCOOH > ClCH 2COOH > CH 3COOH Oxidation The notion of oxidation state is useful in categorizing many organic transformations. 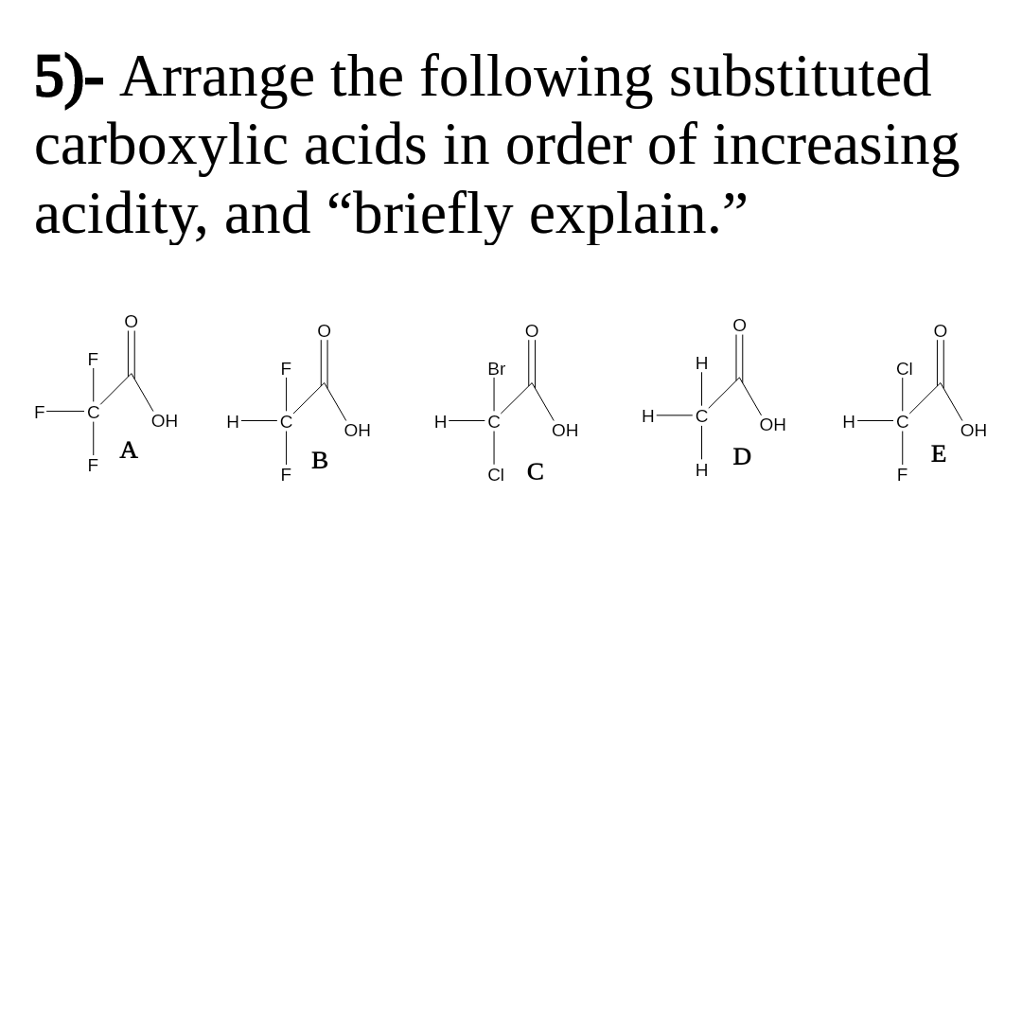 
In the light of this, the following are the orders of a few substituted carboxylic acids. For example, electron withdrawing group disperses the negative charge of the anion and hence makes it more stable causing increase in the acidity of the corresponding acid, on the other hand, electron-releasing group increases the negative charge on the anion and hence makes it less stable causing the decrease in the acidity. Since acidity is due to the resonance stabilization of anion, substituent causing stabilization of anion increases acidity whereas substituent causing destabilization of anion decrease acidity. The acidity of carboxylic acid depends very much on the substituent attached to – COOH group. In the carboxylic acid, these bond lengths are no longer identical. The acidity of a carboxylic acid is due to the resonance stabilization of its anion.īecause of the resonance, both the carbon oxygen bond in the carboxylate anion have identical bond length. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
